Science : Term 3 Unit 3 : Polymer Chemistry
EVALUATION
I. Choose the correct answers
1. The first man-made fibre is _________
a. Nylon
b. Polyester
c. Rayon
d. Cotton
[Answer: (c) Rayon]
2. Which of the following is the strongest?___________
a. Rayon
b. Nylon
c. Acrylic
d. Polyester
[Answer: (b) Nylon]
3. When you place a natural fibre in a flame it____________
a. melts
b. burns
c. gets nothing
d. explodes
[Answer: (b) burns]
4. A synthetic fibre which has similar properties to wool is ____________
a. Nylon
b. Polyester
c. Acrylic
d. PVC
[Answer: (c) Acrylic]
5. A good application of plastic is the use of____________
a. Blood bags
b. Plastic cutlery
c. Plastic straws
d. Plastic carry bag
[Answer: (a) Blood bags]
6. ____________ is a non-biodegradable material
a. Paper
b. A plastic bottle
c. Cotton cloth
d. Wool
[Answer: (b) A plastic bottle]
7. PET is the acronym for ____________
a. Polyester
b. Polyester and terylene
c. Polyethylene terephthalate
d. Polyetheneterylene
[Answer: (c) Polyethylene terephthalate]
II. Fill in The Blanks
1. Raincoat is an example of polyester fabric.
2. Resin codes are used to identify different types of plastics.
3. A polymer is a long chain made up of many repeated small units called monomers.
4. A natural fibre is called plant fibre
5. A natural fibre obtained by boiling cocoons is called silk.
III. True or False
1. A lot of plastic pollutes our environment. Answer: True.
2. Refuse (avoid) is the best way to manage plastic. Answer: True.
3. It is good to wear clothes made of synthetic fibres while cooking. Answer: False.
Correct statement: It is good to wear clothes made of natural fibres while cooking.
4. Degradable plastics break down into tiny pieces called microplastics. Answer: True.
5. Cotton is a natural polymer. Answer: True.
IV. Match the Following
A/ B
Nylon – Thermoplastic
PVC – Thermosetting plastic
Bakelite – Fibre
Teflon – Wood pulp
Rayon – Non-stick cookwares
Answer:
A B
1. Nylon : Fibre
2. PVC : Thermoplastic
3. Bakelite : Thermosetting plastic
4. Teflon : Non-stick cookwares
5. Rayon : Wood pulp
V. Arrange in Correct Sequence
1.Mix water, starch, vinegar and glycerin in a cooking pot.
2. Let the article cool for 24 hours before we use it.
3. Shape material to form a cup or bowl.
4. Continuously mix on medium heat until the liquid turns clear
5. When the liquid begins to bubble it is ready to be taken off the stove.
6. Spread the gel onto aluminium foil and cool.
Answer:
1. Mix water, starch, vinegar and glycerin in a cooking pot.
4. Continuously mix on medium heat until the liquid turns clear.
5. When the liquid begins to bubble it is ready to be taken off the stove.
6. Spread the gel onto aluminium foil and cool.
3. Shape material to form a cup or bowl.
2. Let the article cool for 24 hours before we use it.
VI. Analogy
1.Cotton: natural : Polyester: Synthetic
2. PLA spoon : compostable : Plastic spoon : Disposable
3. Nylon :melts on heating : Silk: Burns on heating
VII. Assertion and Reason
1. A: Vegetable peels buried in the soil disappear within two weeks.
R: Vegetable peels are compostable.
[Answer: (a) Both A and R are true and R is the correct explanation of A]
2. A: It takes a very long time for nylon clothes to breakdown into microfibers but cotton clothes need only six months to decompose.
R: Nylon made out of petrochemicals is non-biodegradable and cotton cloth is biodegradable.
[Answer: (a) Both A and R are true and R is the correct explanation of A]
3. A: It is good to avoid plastics.
R: Plastics end up polluting the environment.
[Answer: (a) Both A and R are true and R is the correct explanation of A]
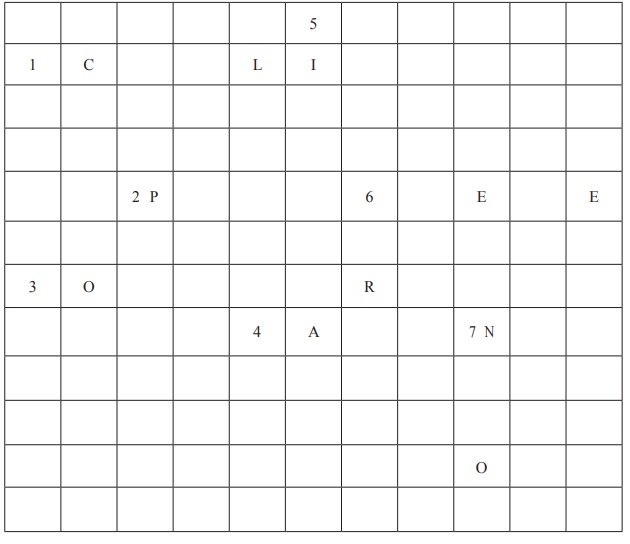
VIII. Crossword
Across
1. Fibre that is used as synthetic wool.
2. A plastic used for making water bottles.
3. A long chain made of small repeatingmonomers.
4. Another name for this semi-synthetic fibre is artificial silk.
Down
5. A type of fibre that is naturally obtained from a cocoon.
6. A synthetic fibre classified as polyester.
7. A polymer used for making rope.

IX. Very Short Answer
1. What is the chemical name of the polymers that make up cotton?
Answer: Cellulose.
2. What gives plastic different qualities and characteristics?
Answer: Different chemicals (additives) are added to plastic to give them various qualities and characteristics.
3. It is not advisable to burn plastic and synthetic fabrics. Why?
Answer: Burning of plastics and synthetic fabrics is not a good solution, as we end up wasting non-renewable resources and produce super toxic chemicals that are difficult to store or dispose safely.
4. A bucket made of plastic does not rust like a bucket made of iron. Why?
Answer:
(i) The reason is due to their chemical composition.
(ii) The bucket is made of plastic which does not react with the oxygen and humidity present in air.
(iii) While the iron reacts with the oxygen, air corrodes to form rust.
5. Why is it better to avoid the use of plastic products?
Answer:
(i) Plastics do not decompose by natural processes and action of bacteria and are therefore not biodegradable.
(ii) A lot of the plastic produced globally is designed to be used only once and thrown away, creating a large amount of plastic waste.
(iii) Plastic waste ends up being recycled, incinerated, landfilled, dumped or ends up littering our environment.
(iv) So, it is better to avoid the use of plastic products.
6. Give two examples of thermosetting plastics.
Answer: Bakelite, Melamine.
7. What is the 5 R principle?
Answer: Plastic disposal is the 5 R principle, Refuse, Reduce, Reuse, Recycle and Recover is called as 5 R principle.
X. Short Answer
1. What does the term biodegradable mean?
Answer: A material that gets decomposed through natural processes and action by bacteria is called biodegradable.
2. What kind of fabric is suitable to dress-up and play in the summer?
Answer:
(i) In summer it is better to wear clothing that is made out of cotton materials rather than synthetic.
(ii) This is because most synthetic fibres absorb very little moisture and do not allow air circulation making them hot and uncomfortable to wear.
3. How do plastics impact animals and the environment?
Answer:
(i) The increase in the use of plastics, particularly the one-time use and throw away plasticshas serious impacts on the environment, animals and our health.
(ii) We have seen garbage dumps with different plastics. One big problem with plastics is that they do not decompose or biodegrade.
(iii) This leads to large amounts of waste that will not disappear and end up accumulating and polluting the environment.
(iv) Many animals confuse plastic for food and eat it by accident. When leftover food is thrown away it is often packed in plastic. Animals smell the leftover food and eat the plastic by accident.
XI. Long Answer
1. List the advantages and disadvantages of synthetic fibres.
Answer:
Advantages and Disadvantages of synthetic fibres.

Advantages of synthetic fibres
1. Do not wrinkle easily and they keep their colour and brightness for a much longer time than natural fibres such as cotton.
2. Using synthetic fibres such as nylon, is that they are stronger than many natural fibres such as silk or wool.
3. These fibres are strong and elastic which gives it the properties to bounce.
Disadvantages of synthetic fibres
1. synthetic fibres such as polyester is that they are not heat resistant and catch fire easily.
2. Most synthetic fibres absorb very little moisture and do not allow air circulation making them hot and uncomfortable to wear.
3. Synthetic fibres are made out of petrochemicals and last in the environment for a very long time. It break down into very small pieces called microplastics which cause pollution to soil and water bodies such as rivers, lakes and oceans.
2. Suggest safe methods of disposing plastics.
Answer: Refuse (Avoid):
(i) The best thing to do is to avoid using plastic products.
(ii) One-time use throw away plastics can often be avoided.
Reduce : Reducing the amount of plastic we use is important.
Reuse : If possible products made of plastics can be used again and again.
Recycle:
(i) It is better to recycle plastic waste.
(ii) Separating plastic waste (based on the resin code) and making sure it gets recycled is good as it turns waste materials into something new.
(iii) Then it will not be thrown away in landfills, open dumps or ending up as litter in the environment.
Recover (Compost and Incinerate): Solid waste can be converted into resources such as electricity and compost through thermal and biological means.
XII. HOTS
1. The Tamil Nadu Government has banned the use of one-time use throwaway plastics. Why do you think this is important?
Answer: It is important to reduce the negative consequences of plastics on the environment.
2. A plastic bag dumped in the soil stays without breaking down for 500 years. If a new generation starts in every 30 years, how many generations would it take to see the plastic bag finally broken down?
Answer: It would take 16 to 17 generations to see the plastic bag finally broken down.
XIII.
i) Fill in the blanks
Answer:
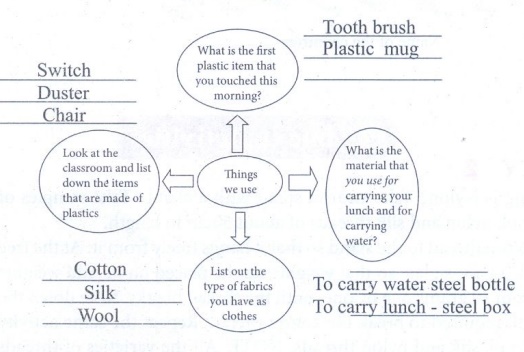
ii. Look at the following picture and explain what is happening.

Answer: A lot of one-time use plastic such as polythene bags and food packaging that are thrown away are responsible for littering the environment and clogging drains. Standing water breeds mosquitoes that can spread diseases such as malaria dengue and chickungunya and also lead to flooding.
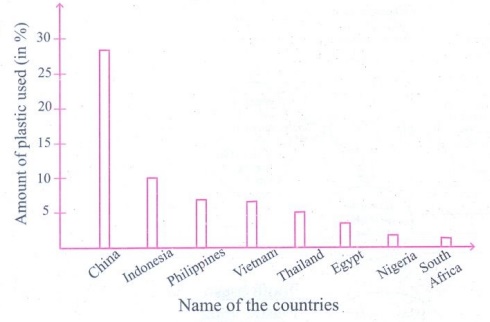
iii. Read the following information and convert them into a graph to compare the countries and the amount of plastic they use.
China contributes the highest share – that is around 28%, of the total plastic used globally. Indonesia uses 10%, both the Philippines and Vietnam use 6% each; Thailand uses 3.2%, Egypt 3%, Nigeria 2.7% and South Africa 2%.
Answer:
Student Activities
ACTIVITY 1
Compare the following things. Ice cubes and a polythene bag. In both the materials, there are large number molecules combined together. Are both polymers?

Nylon is very strong and can be used for rock climbing!
Nylon is a plastic polymer made of chemical units called polyamides. olyamides are made with monomers – hexamethylenediamine and adipic acid. Solid chips of these polyamides are melted and forced through a heated spinneret which has very, very tiny holes.

ACTIVITY 2
How Strong is Nylon?
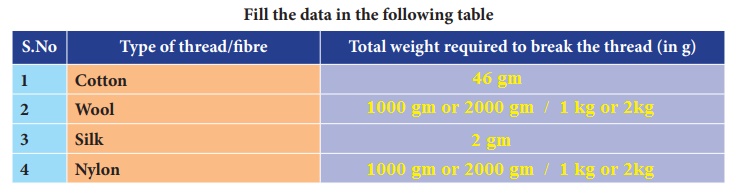
Take an iron stand with a clamp. Take samples of cotton, wool, nylon and silk threads of about 50cm in length.
First tie cotton thread to the stand so that it hangs freely from it. At the free end, attach a CD as plate so that weights can be placed on it. Add weights starting from 10 grams one by one, untilthe thread breaks. Note down the total weight required to break the cotton thread. Repeat the same activity with the wool, silk and nylon threads.
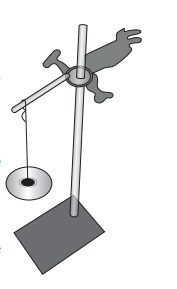
Iron clamp with a hold weights
NOTE: All the varieties of threads should be of same thickness.
Arrange the threads in the order of increasing strength.
Answer: Cotton, Wool, Silk, Nylon.
What do you infer from the above activity?
Answer: Nylon thread is the strongest thread.
Which type of fibre is the strongest?
Answer: Nylon.
Which type of fibre is the weakest?
Answer: Cotton.
ACTIVITY 3
Identify The Fibre : Let us do an activity. Look at the images below and identify and write down the name of the different types of fibre observed.

Answer:
1. Jute – Natural
2. cotton – Natural
3. silk saree – Natural
4. Rayon material – Synthetic
5. wool – Natural
6. polyester – Synthetic
ACTIVITY 4
Synthetic or Natural Fibres : The teacher can give the learner a piece of each and every type of fibre. The learner can feel the fibre and write down the name of the fibre and state whether it is natural or synthetic fibre.

1. Jute – Natural
2. Cotton – Natural
3. Rayon – Synthetic
4. Wool – Natural
5. Polyester – Synthetic
We have done four activities so far. Which activity helped you better to identify the type of fibre? Answer: Activity 3, 4
(Both the use of familiar images as well as touch can help us to identify the different types of fibres. Right!) We have learned about fabrics such as polyester, nylon and acrylic and their common uses. These synthetic fibres are polymers which we will learn about later in this chapter.
ACTIVITY 5
Burning A Piece of natural fibre and a synthetic Fibre
Note: This activity should be performedonly by the teacher.
Take a piece of cotton cloth and a piece of polyester cloth. Both the pieces of cloth can be of same size (2cm x 2cm is enough). Hold the pieces of cloth using tongsto protect yourself from the flame and heat. Burn both the pieces of cloth one by one and see what happens when they burn.
What do you observe while the cotton cloth burns?
Answer: The cotton cloth burns completely. Does not melt and has the odour of burning paper.
What do you observe while the polyester cloth burns?
Answer: Polyester cloth melts on burning and giving off black smoke.
You must have noticed that natural (cotton) fibresburn very differentlycompared tosynthetic (polyester) fibres.

The cotton cloth will burn when it is held in flame. On the other hand, the synthetic polyester cloth will melt on burning. This is one of the disadvantages of wearing clothes madeout of synthetic fibres. If a synthetic cloth comes into contact with fire the fabric melts and sticks to the body causing severe burns. Therefore, one should not wear synthetic clothes while cooking or working in a laboratory.
ACTIVITY 6
Wetting A Cotton Cloth And An Umbrella Cloth
We use an umbrella on rainy days. What kind of umbrella do we use?
Answer: Nylon or Polyester
Can you use an umbrella made of cotton?
Let us do an activity to see why we do not use a cotton umbrella to protect ourselves from the rain. Take a piece of cotton cloth of approximately 10 cm X 10 cm size and a piece of nylon or polyester cloth 10 cm X 10 cm in size from an old unusable / untorn umbrella. Ask four students to hold the four corners of the piece of cotton cloth and pour a glass of water over it. Then ask four students to hold the piece of umbrella cloth and pour a glass of water over it. Compare the effect of water on the piece of cotton cloth and umbrella cloth and record your observation in the notebook.
Which of these fabrics allows water to pass through?
Answer: Cotton cloth allows water to pass through whereas umbrella cloth does not allow water to pass through.
Cotton cloth or Umbrella cloth (nylon or polyester) Now ask the students to put both the pieces of cloth in the hot sun to dry. Which of these fabrics dries the fastest? The cotton cloth or the umbrella cloth?
Answer: Umbrella cloth dries the fastest.
Synthetic fibres are made from the byproducts of processing petroleum oil and gas. You will learn about fractional distillation of petroleum in your higher classes.

It is estimated that every year we use a trillion plastic carry bags (2 million a minute) around the world and out of which only 1 to 3% are recycled.

Plastic has been around for less than 200 years. Edmund Alexander Parkes was the creator of the first plastic called ‘Parkesine’

ACTIVITY 7
Right and wrong application of plastics
Look at the list of eight plastic items. Decide which fourplastic itemsare used for the right application and which four are used for the wrong application by filling in the chart below:
Plastic items: straws, helmets, cutlery, thin carry bags, syringes, electrical wires,tea cups and blood bags

Right application : Wrong application
Helmets : Straws
Syringes : Thin carry bags
Electrical wires : Cutlery
Blood bags : Tea cups
ACTIVITY 8
Identify the different types of plastics
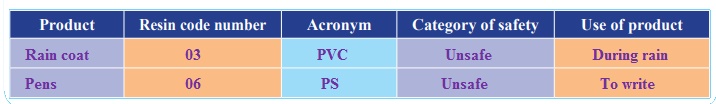
Collect different kinds of plastic products and look carefully for the resin code and/or acronym on them. With the help of the resin code chart,mark the resin code number, acronym, if you think it is a safer, unsafe or questionable (when you cannot find the resin code on the article) type of plastic. What resin codes do you find? Is the resin code safer,unsafe or questionable?
A recipe for PLA a compostable plastic!
What you need
i. 1 tablespoon of corn starch
ii. 1 teaspoon of vegetable glycerin (available at the pharmacy)
iii. 1 teaspoon of vinegar (5% acidity)
iv. 4 Tablespoons of water.
v. Cooking spoon
vi. Cooking pot
vii. Stove
viii. Aluminium foil
Method
Mix the water with the starch in a cooking pot. Add the vinegar and the glycerin. Mix all the ingredients on medium heat. Make sure you continuously stir. The mixture should turn from liquid white into a clear gel. When it begins to bubble, then it is ready and should be taken off the stove.
Spread the gel onto the aluminium foil. Let it cool down for one hour. You can then shape the material to form a cup or bowl. Let the article you made cool for another 24 hours before you try and use it.
