Science : Chapter 14 : Acids and Bases
TEXTBOOK EXERCISES
I. Choose the best answer.
1. Acids are ________ in taste.
a) sour
b) sweet
c) bitter
d) salty
[Answer: (a) sour]
2. Aqueous solutions of _________ conduct electricity.
a) acid
b) base
c) salt
d) All of these
[Answer: All the above]
3. In acidic solutions blue litmus changes into ______ colour.
a) blue
b) green
c) red
d) white
[Answer: (c) red]
4. Base is a substance that gives _____ on dissolving in water.
a) OH−
b) H+
c) OH
d) H
[Answer: (a) OH−]
5. Sodium hydroxide is a ____________
a) acid
b) base
c) oxide
d) alkali
[Answer: (d) alkali]
6. Red ant sting contains __________
a) acetic acid
b) sulphuric acid
c) oxalic acid
d) formic acid
[Answer: (d) formic acid]
7. Magnesium hydroxides are used for treating _______
a) acidity
b) head pain
c) teeth decay
d) None of these
[Answer: (a) acidity]
8. Acid mixed with base forms ________
a) salt and water
b) salt
c) water
d) No reaction
[Answer: (a) salt and water]
9. We brush our teeth with tooth paste because it is __________ in nature.
a) basic
b) acidic
c) Both a and b
d) None of these
[Answer: (a) basic]
10. In basic solution turmeric indicator paper changes from yellow to __________
a) blue
b) green
c) yellow
d) red
[Answer: (d) red]
II. Fill in the blanks.
1. Benzoic acids are used for preservation of food.
2. The word sour refers to acidus in Latin.
3. Bases are bitter in taste.
4. Chemical formula of calcium oxide is CaO.
5. Wasp sting contains alkaline substance.
6. Turmeric is used as a indicator.
7. In acidic solution the colour of the hibiscus indicator paper will change to deep pink or deep red.
III. State true or false. If false, correct the statement.
1. Most of the acids are not soluble in water. [Answer: True.]
2. Acids are bitter in taste. [Answer: False.]
Correct statement: Acids are sour in taste. Bases are bitter in taste.
3. Bases are soapy to touch when they are dry. [Answer: False.]
Correct statement: Bases are soapy to touch only in aqueous media, not in dry nature.
4. Acids are corrosive in nature. [Answer: True.]
5. All bases are alkalis. [Answer: False.]
Correct statement: All alkalis are base, but all bases are not alkalis.
6. Hibiscus flower is an example for natural indicator. [Answer: True.]
IV. Answer briefly.
1. Acid – Define.
Answer: A substance which contains one or more replaceable hydrogen atoms.
2. Write any four physical properties of acids.
(i) Acids are sour in taste.
(ii) They are corrosive in nature. Strong acids can spoil substances like human skin, clothes and paper.
(iii) Generally acids exist in liquid state but few acids exist in solid state too. E.g. Benzoic acid.
(iv) Acids are colourless.
(v) Acids change the colour of the indicators. Blue litmus paper turns red and methyl orange turns pink when treated with acids.
3. What are the similarities between acids and bases?
Answer:
(i) They are corrosive in nature.
(ii) They undergo ionization in aqueous solution.
(iii) They conduct electricity in aqueous solution.
(iv) They undergo neutralization reaction.
4. State the difference between acids and bases.
Answer: Difference between acids and bases
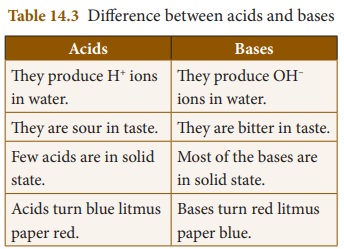
Acids
They produce H+ ions in water.
They are sour in taste.
Few acids are in solid state.
Acids turn blue litmus paper red.
Bases
They produce OH− ions in water.
They are bitter in taste.
Most of the bases are in solid state.
Bases turn red litmus paper blue.
5. What is an indicator?
Answer: An indicator or acid – base indicator is a chemical substance which indicates the acidic or basic nature of a solution by suitable colour change.
6. What is a neutralization reaction?
Answer: Neutralization is a chemical reaction in which an acid and a base react with each other to form water and salt.
7. Write any four physical properties of base.
Answer:
(i) Bases generally exist in solid state but some bases exist in liquid state also.
E.g. Ammonium hydroxide, calcium hydroxide.
(ii) Bases give soapy touch only in aqueous media not in dry nature.
(iii) Bases are bitter in taste.
(iv) Bases are corrosive in nature. When come in contact with the skin frequently they form painful blisters.
(v) Bases also change the colour of the indicators. Red litmus paper turns blue when treated with bases. Similarly, they turn methyl orange yellow and phenolphthalein pink.
V. Answer in detail
1. What are the uses of acids?
Answer:
(i)Hydrochloric acid present in our stomach helps in the digestion of foodstuff.
(ii) Vinegar (acetic acid) is used to preserve food materials.
(iii) Benzoic acid is also used to preserve food materials like pickles.
(iv) Sodium or potassium salts of higher fatty acids are used to make washing and bathing soaps.
(v) Sulphuric acid is called the king of chemicals. It is an effective dehydrating agent.
It is used in various industries to make detergents, paints, fertilizers and many more chemicals.
(vi) Hydrochloric acid, Nitric acid and Sulphuric acid are important laboratory reagents,
(vii) Cells of all living organisms contain the fundamental nuclear material called nucleic acids. Animals have deoxyribo nucleic acid (DNA) whereas plants contain ribo nucleic acid (RNA).
2. What are the uses of bases?
Answer:
(i) Potassium hydroxide is used to make bathing soaps.
(ii) Sodium hydroxide is used to make washing soaps.
(iii) Sodium hydroxide is also used in paper industries, textile industries and in the preparation of medicines.
(iv) Calcium hydroxide is used for white washing.
(v) Aluminum hydroxide and magnesium hydroxides are used in antacids to cure acidity problems.
(vi) Ammonium hydroxide is used to manufacture fertilizers, nylon, plastics and rubber.
3. Explain the neutralization reactions in our daily life.
Answer:
Ant bite:
(i) Whenever bees or red ants bite they inject an acid called formic acid.
(ii) These acids cause burning sensation and pain.
(iii) To suppress the pain, a suitable base in the form of calcium hydroxide (readily available at home) is applied to neutralise the formic acid.
Wasp bite :
(i) When we are bitten by wasp, we feel the burning sensation and pain. It is due to an alkaline substance injected by the insect.
(ii) To neutralise the alkalinity, we use vinegar which is an acid.
Tooth decay :
(i) The bacteria present in our mouth decompose the food particles stuck in the gaps between our teeth thereby causing acid formation which leads to tooth decay.
(ii) When we brush with tooth powder or tooth paste containing weak bases, the acid gets neutralized.
Acidity :
(i) Excessive production of hydrochloric acid in our stomach causes ulcer in stomach and food pipe.
(ii) In order to neutralize, antacids which are nothing but weak bases like aluminum and magnesium hydroxides are used.
Agriculture :
Farmers add lime fertilisers such as powdered lime (CaO), limestone (CaCO3) or ashes of burnt wood to the soil to neutralise the acidity.
Industries :
Effluents from the industries contain acids such as sulphuric acid. It is treated by adding lime to neutralise it before it is discharged into rivers and streams.
4. How will you prepare natural indicator from turmeric powder.
Answer:
(i) Turmeric indicator is one of the natural indicator.
(ii) By adding small amount of water to turmeric powder, a paste is prepared.
(iii) This is applied on a blotting paper or filter paper and dried.
(iv) These strips are used as indicators to find the nature of the solution.
(v) In acidic solution, turmeric indicator paper has no change in colour.
(vi) That means, it remains yellow. In basic solution, the colour changes from yellow to red.
VI. Higher Order Thinking Questions.
1. Vinu and Priyan take their lunch at school. Vinu eats lemon rice and Priyan eats curd rice. Both lemon rice and curd rice are sour in taste. What is the reason?
Answer:
(i) Curd contains lactic acid. The lactic acid makes curd rice sour in taste.
(ii) Lemon juice contains citric acid. The citric acid makes lemon rice sour in taste.
(iii) Generally acids are sour in taste.
2. Heshna and Keerthi are friends. Keerthi’s teeth are white without caries, but Heshna has teeth with caries. Why? How is it formed?
Answer:
(i) Caries is caused by the action of acids on the enamel surface.
(ii) The acid is produced when sugar in foods or drinks react with bacteria present on the tooth surface.
(iii) Heshna has not cleaned her teeth well after sipping sugary drinks and snacking.
(iv) She has to brush after meals and before bed.
Student Activities
Activity 1
Take a clean test tube with holder and pour some dilute hydrochloric acid. Add few pieces of magnesium ribbon slowly. What do you observe? Now show a burning match stick near the mouth of the test tube. Do you hear any sound? The gas burns with a pop sound. From this it is observed that hydrogen gas is formed due to the reaction between acid and metal (Do it under the supervision of the teacher).
Copper or brass cooking vessels are coated with tin metal (eyam). If it is not coated the organic acids present in the food materialswill react with copper and make the food poisonous. The tin isolates the vessel from the action of acids and prevents food poisoning.
Answer: The gas burns with a pop sound. From this it is observed that hydrogen gas has been formed due to the reaction between acid and metal.
Activity 2
Take some lemon juice in a tumbler and add baking soda slowly. What do you see? What do you infer from this?
Answer:
Inference : When lemon juice is mixed with baking soda, the new product CO2 is formed with water and salt.
Activity 3
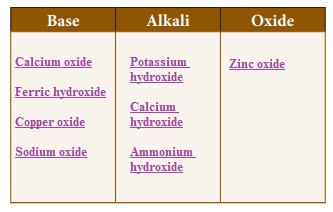
Classify the following substances.Sodium oxide, Potassium hydroxide, Calcium oxide, Copper oxide, Calcium hydroxide, Ammonium hydroxide, Ferric hydroxide, Zinc oxide
Activity 4
Take a white cloth with turmeric powder stain. Wash the cloth with washing soap. Do you observe any change in the colour? Why?
Answer: Yes, the colour changes from yellow to red, because soapy solution is a base.
Activity 5
Take a small beet root vegetable and cut it into pieces. Boil them in hot water and filter the extract. Take two test tubes. Take sodium hydroxide solution in one test tube and vinegar or lemon juice in another test tube. Add beet root extract slowly. Observe the colour change. What do you infer?
Answer:
(i) Observation : When beetroot juice is added with sodium hydroxide solution it turns into greenish yellow shows that NaOH – is a base.
(ii) When it is added with lemon juice, the colour of beetroot juice remains same shows that lemon juice is acidic.
Activity 6
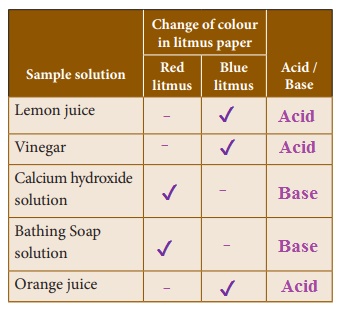
Find out the nature of the solution.
Swedish chemist Svante Arrheniu proposed a theory on acids. According to him,an acid is a substance which furnishes H+ ions or H3O+ ions in aqueous solution.

We feel hungry due to thecorrosive action of hydrochloric acid on the inner lining of the stomach. When the level of hydrochloric acidgoes higher, it causes ulcer.
Pickles remain in good condition for long time because they contain vinegar (acetic acid) or benzoic acid.

Sodium carbonate (Na2CO3) is commercially called as washing soda. Similarly sodium bicarbonate (NaHCO3) is commercially called as baking soda. Caustic soda is sodium hydroxide (NaOH) and caustic potash is potassium hydroxide (KOH).

Tamil